How to Fill Lead Acid Battery? Lead Acid Battery Watering Tips.Can You Mix Lead-acid and Lithium Batteries?.Similar principles are used to make lead-acid batteries, but the substance is different. Positive and negative ions from the lithium material are discharged between electrodes in lithium-ion batteries to operate. How Do Lithium-ion and Lead Acid Batteries Work?īoth batteries function by electrochemically storing a charge and releasing electrons. Lead-acid batteries are made with the metal lead, while lithium-ion batteries are made with the metal lithium, as their names would suggest. Lead acid and lithium-ion are two of the most popular battery chemistry types. Lead Acid Batteries Overviewįor solar energy systems, battery storage is a feature that is increasingly in demand. Can Lead-acid Battery Last for 10 Years?.Why Are Lead-acid Batteries Better Than Lithium?.Do Lead-acid Batteries Last Longer Than Lithium-ion?.Conclusion: Lead Acid Battery Vs Lithium-ion.When Should You Install a Lead Acid Battery Vs.How Do Lithium-ion and Lead Acid Batteries Work?.The recycling process is often regulated by governments and people are paid to properly dispose of their batteries. Because of this, it is important to properly recycle lead-acid batteries. Some lead compounds are extremely toxic and could cause lasting health problems. Both lead and sulfuric acid can contaminate soil and groundwater. Excessive charging with high voltages can lead to gases building up and the battery could explode.įrom an environmental standpoint, lead-acid batteries are a problem. It is important to charge them with optimal voltage. The main downsides of lead-acid batteries are that they charge slowly, their cycle life is limited and they need regular maintenance. 
But lead-acid batteries are still used in cheaper models to bring down the cost. For example, lithium-ion batteries have a much higher energy density and therefore are suited for use in places where the size of the battery is important such as electric scooters. Because of this, lead-acid batteries are widely used even in places where newer technologies would suit better. Lead-acid batteries have a large power-to-weight ratio which makes them good for supplying high surge currents. Deep-cycle batteries are used to give continuous power to electric vehicles such as wheelchairs and scooters, and also for power storage. 
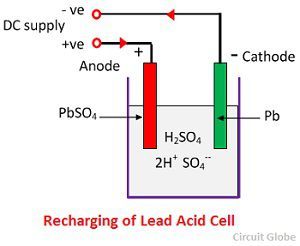
These batteries are designed to have a good capacity and high cycle count. Starting batteries are not designed for repeated deep discharge and charge cycles.ĭeep-cycle batteries have thick electrodes to improve cycling performance. When the engine is running, the alternator charges the battery and holds the charge at the optimal level. They are designed to be used for starting an engine. These batteries give out a high burst of current at once. Starting batteries have a large number of thin electrodes to maximise surface area. Two main types of lead-acid batteriesīy design, there are two types of lead-acid batteries: starting batteries and deep-cycle batteries. This forces the reverse reaction to take place. When charged, the charger creates an excess of negative electrons on the anode and hydrogen ions are attracted to it. As a result of the total reaction, both electrodes are covered in lead sulfate (PbSO 4) and the electrolyte becomes primarily water. On the cathode, the PbO 2 reacts with sulfuric acid. When using the battery, the lead reacts with the sulfuric acid releasing electrons and hydrogen ions (H +) that move from anode to cathode. The electrolyte is a concentrated sulfuric acid solution (H 2SO 4). When fully charged, the anode consists mainly of lead (Pb) and the cathode of lead dioxide (PbO 2). How do lead-acid batteries work?Ī lead-acid battery consists of negative electrodes (anodes), positive electrodes (cathodes), separators between them, and electrolyte. In addition, it is also used for storage in backup power supplies. It is commonly used as an automotive battery for starting the engine but also as the main energy source for electric vehicles such as motorised wheelchairs, golf carts and electric scooters. Lead-acid battery is the first rechargeable battery ever created.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
